Translate this page into:
Gastroesophageal reflux disease in bronchial asthma: What we need to know?

-
Received: ,
Accepted: ,
How to cite this article: Patil S, Gondhali GV, Choudhari SR. Gastroesophageal reflux disease in bronchial asthma: What we need to know? Glob J Health Sci Res 2023;1:110-5.
Abstract
Gastroesophageal reflux disease (GERD) is most common disease with variable presentation from asymptomatic to frank symptomatic disease having impact on various respiratory disease such as cough, bronchitis, bronchial asthma, chronic obstructive pulmonary disease, and interstitial lung diseases. GERD is defined by retrograde reflux of gastric contents to the esophagus leading to various signs and symptoms that range from heartburn/ regurgitation to the development of extraesophageal respiratory syndromes like asthma. Bronchial asthma and GERD are more common diseases and usually reported more frequently as “two side sides of same coin.” GERD has been associated with a causal link for asthma, worsening of asthma control, one of the causes for refractory asthma, and a main factor in asthma cases with recurrent asthma exacerbation. Asthma itself and medicines used for the treatment of asthma are associated with GERD, worsening of GERD and one of the causes for refractory GERD. However, asthmatics have been found to have a much greater prevalence of GERD symptoms than the general population. There remains debate regarding the underlying physiologic mechanism of this relationship and whether treatment of GERD actually translates into improved asthma outcomes. The clinical prevalence of the two conditions in many patients cannot be ignored, and multiple theories highlighting plausible pathogenic explanations for these associations have been described. The treatment of symptomatic GERD in patients with asthma is important to control the underlying GERD and should be undertaken based on current guidelines for the treatment of the GERD.
Keywords
Bronchial asthma
Cough
Gastroesophageal reflux disease
Proton pump inhibitors
Chronic obstructive pulmonary disease
INTRODUCTION
Gastroesophageal reflux disease (GERD) is a prevalent condition defined by a chronic retrograde movement of gastric content to the esophagus leading to a wide range of symptoms and complications in patients. Risk factors such as female gender, obesity (body mass index >30), and advancing age have all been associated with an increase in GERD-related symptoms. The pathophysiology of GERD is a complex topic that involves several mechanisms that impair the protective capacity of the esophagus to overcome the reflux of acid, pepsin, duodenal bolus, and pancreatic enzymes. The mechanisms implicated include motor anomalies, anatomical anomalies, and impaired resistance of the mucosa. GERD-related symptoms arise due to lesions of the mucosa lining the esophagus and may manifest as esophageal (regurgitation and heartburn) or extraesophageal (chronic cough, asthma, dental erosions, laryngitis, and non-cardiac chest pain) findings.[1-3] GERD clinical prevalence in asthmatic patients is widely variable, and it is estimated to be around 30–50%.
In 1966, a link was discovered between GERD and respiratory diseases, implicating GERD in the pathogenesis of asthma. GERD is frequently cited in asthmatic patients. A systematic review of 28 studies determined the prevalence of gastroesophageal reflux (GER) symptoms in asthmatic patients to be 59.2% compared to 38.1% in controls.[4,5]
The explanation behind bronchoconstriction due to acid reflux can be explained by three postulated mechanisms: Increased bronchial reactivity, micro-aspiration of acid and other gastric contents into the upper airways, and increased vagal tone. Other authors have suggested that the association between GERD and asthma can be explained by the “reflux theory,” otherwise known as a direct mechanism and the “reflex theory” as an indirect mechanism.[6]
DISCUSSION
Possible link in asthma and GERD
There have been several theories put forth in an attempt to explain how GERD and asthma may be linked in a causal fashion.[7]
Asthmatic patients often have lung hyperinflation. Descent of the diaphragm in the setting of lung hyperinflation and increased work of breathing increases the pressure gradient between the abdomen and chest and may cause the lower esophageal sphincter (LES) to herniate into the chest where its barrier function is impaired. This could therefore allow more reflux of gastric contents among asthmatics with hyperinflation.[8]
Some asthma medications aimed at reducing hyperinflation may promote acid reflux. Both beta-agonists and theophylline may decrease LES tone, which again potentially could foster acid reflux. One study of theophylline showed that symptoms of GERD increased by 170% in the theophylline group compared with placebo. Beta-agonists have been shown to cause reductions of LES tone in a dose-dependent fashion. This suggests the potential for a vicious cycle of GERD-induced asthma symptoms resulting in increased use of bronchodilators, which in turn promotes more GERD.[9]
There are also some GERD-related pathogenic factors which could worsen asthma.[7]
The parasympathetic nervous system, specifically the vagus nerve, highly innervates both the esophagus and the tracheobronchial tree and may be a common pathogenic pathway for GERD and asthma. In animal studies, instillation of acid into the esophagus was shown to increase respiratory resistance with effects ablated by bilateral vagotomy
In humans, instillation of acid in the esophagus has been shown to decrease peak expiratory flow rates and increase overall airway resistance. Both of these consequences are blunted by pretreatment with atropine. In contrast, others have found no relationship between the instillation of acid and methacholine reactivity or lung function. Some data seem to suggest that acid refluxed from the stomach into the esophagus may “prime” the lung to have subsequent and more severe episodes of bronchospasm when exposed to another trigger[10]
Another question raised has been whether GERD-induced aspiration or microaspiration could worsen asthma, as tracheal microaspiration of acid has been found to be a very potent stimulus for bronchospasm. In addition, microaspiration may trigger bronchospasm indirectly by inducing chronic inflammatory changes which subsequently can lead to increased airway reactivity. One study showed that esophageal acid led to significantly increased levels of Substance P, a tachykinin that leads to smooth muscle contraction and increased vascular permeability. Acid provocation has also been shown to lead to increased lung resistance in a dose-dependent fashion which is mediated by release of tachykinins from peripheral nerves. Aspiration of acid may also cause injury directly to the epithelial lining of the upper airway which has been shown to result in a release of cytokines and increased inflammation.[11]
“Reflux theory” and “Reflex theory”
The cause-effect relationship between GERD and asthma is difficult to establish since it has been proven that either disease process can trigger the development of each other. GERD-Induced asthma should be suspected in all patients who present at an adult age with new-onset asthma symptoms, poor asthma control even with proper medication, and heartburn or regurgitation preceding asthmatic events. The symptoms of GER, such as heartburn and regurgitation, affect a large percentage of asthma patients, and the absence of traditional symptoms does not rule out physiologic acid reflux in those who have a chronic cough. A systematic review conducted in 2007 by Havemann et al. discovered that the average prevalence of GERD symptoms in asthma patients was 59.2%. However, the prevalence of asthma in patients with GERD was estimated to be 4.6%.[5] The complex pathophysiology explaining the correlation between asthma and GERD is still a heavily debated topic. However, two proposed mechanisms have been studied and are widely accepted; a “reflux theory” and a “reflex theory.” [Figure 1] is showing asthma and GERD overlapping pathophysiology and link between two diseases.
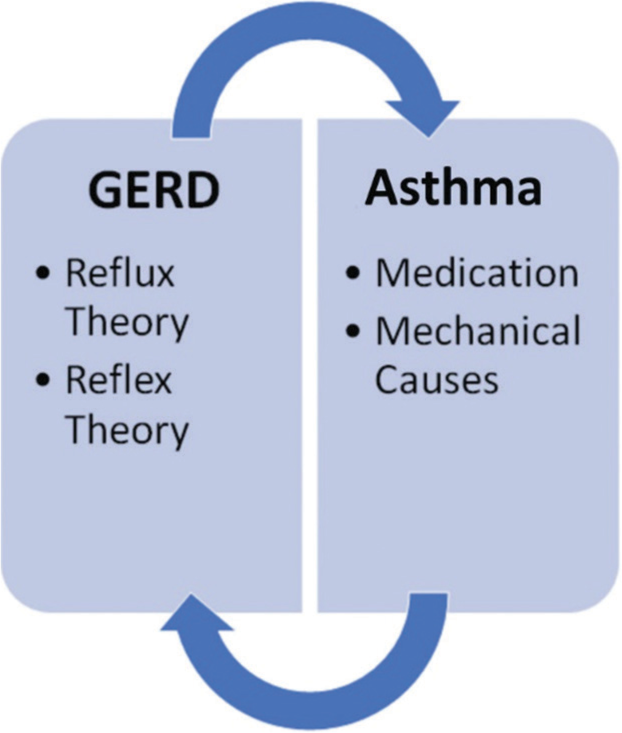
- Proposed mechanisms between gastroesophageal reflux disease and asthma.[2]
The reflux theory[2]
This theory refers to a direct mechanism by which gastroduodenal reflux (acid, pepsin, bile acids, and pancreatic enzymes) enter the esophagus and subsequently leads to microaspiration into the lungs and other parts of the pulmonary tree. The aspiration of these contents can result in stimulation of the pharynx or larynx, consequently resulting in extraesophageal symptoms such as tracheal or bronchial cough reflex. The chronic inflammation of lung tissue may lead to airway obstruction, poor gas exchange, acute lung injury, and severe acute respiratory distress syndrome. These mechanisms consequently induce the liberation of proinflammatory cytokines from T-helper Type 2 cells, leading to an increase in airflow resistance and inflammation. Bronchoconstriction is induced by direct HCl intra-esophageal instillation through muscarinic (M3) receptors, which release acetylcholine. This contributes to airway inflammation and the stimulation of smooth muscle contractions in the airways. This contributes to airway inflammation and the stimulation of smooth muscle contractions in the airways. Infiltrates of macrophages, neutrophils, eosinophils, and lymphocytes are detected histologically in GERD-induced airway inflammation.[12-14]
The reflex theory[2]
The reflex theory refers to a method otherwise known as an indirect process by which distal esophageal reflux induces stimulation of the vagus nerve leading to bronchoconstriction. This method operates on the basis that the esophagus and tracheobronchial tree share an embryological origin and would therefore share similar innervation. Consequently, this shared innervation could explain why stimuli in the distal esophagus led to extraesophageal symptoms through vagus nerve reflexes.[15] A study conducted by Harding et al. in 1999 sought to examine the prevalence and severity of GERD in asthmatics with and without reflux symptoms as well as the correlation of respiratory symptoms with intra-esophageal acid. A total 82% of asthmatics had underlying reflux symptoms, 72% asthmatics with reflux symptoms had abnormal 24-h Esophageal pH tests versus 30% asthmatics without reflux symptoms. Among asthmatics with GER, esophageal acid was linked to respiratory symptoms in 78.8% cases.[15]
GERD and asthma exacerbations
GERD is highly prevalent and often coexists with asthma exacerbation. The meta-analysis summarizes the association between GERD and asthma exacerbation, two highly prevalent diseases that often coexist, but no concise evidence about their association is available in the literature. Our findings show that patients with GERD have 27% increased odds of suffering asthma exacerbations compared with asthma patients without GERD and that the association is more pronounced in patients with pediatric age. The occurrence of asthma exacerbation is associated with the copresence of GERD disease in people with asthma.[16]
Establishing a relationship between GERD and asthma exacerbations is complicated by the fact that GERD is often “silent,” and as a result, questionnaires and clinical history taking are unreliable diagnostic methods. The most reliable method used in the studies included in the meta-analysis was 24-h pH probe monitoring, and the four studies that employed this technique in at least some patients reported an association between GERD and asthma exacerbation which almost double the magnitude of that estimated for all studies (50% vs. 27%).[16,17]
Treatment of GERD in asthma
Considering that a relationship between GERD and asthma has been shown, it is reasonable to assume that treating GERD in asthmatics may be beneficial. In roughly 70% of patients, anti-reflux medication improves asthma-related symptoms (wheezing, coughing, dyspnea, and chest discomfort). Two pharmacologic agents have been described in the literature as an effective therapy for GERD-related symptoms in asthmatic patients; histamine receptor antagonists; and proton pump inhibitors (PPIs).[2,18]
Treatment with antireflux therapy did not consistently improve any one measure of pulmonary function in patients with asthma, but some studies did report objective improvement in single measurements of lung function such as forced expiratory volume in one second (FEV1), morning peak expiratory flow rates (PEF), and evening PEF. The variability in reporting of asthma symptoms in the included studies prevented detailed analysis of any parameter except for nocturnal asthma symptoms which were noted to be improved by some authors in patients treated with histamine antagonists or omeprazole. However, others failed to show improvement in nocturnal asthma symptoms with GERD therapy.
H2 receptor antagonists
Inhibition of the H2 receptor on parietal cells in the stomach mucosa by receptor antagonists like cimetidine or ranitidine leads to reduced gastric acid secretion. The majority of studies indicate an improvement in reflux symptoms and, in some cases, recovery from nocturnal asthmatic symptoms. Ranitidine 300 mg was given as a daily dose (unless weight <40 kg, in which case 150 mg twice daily was used). In individuals with pathological GERD, ranitidine showed a small (30%) but statistically significant decrease in nocturnal asthma symptoms. In summary, medical therapy with ranitidine did not improve pulmonary function, medication requirements, survival, or improvement of asthma symptoms.
PPIs
PPIs are an innocuous and relatively well-tolerated family of medications that directly inhibit the proton pump (H+/K+ ATPase) of the parietal cells in the stomach leading to the suppression of acid. PPIs are referred to as the current gold standard in the treatment of GERD because they provide a demonstrable advantage over histamine blockers in terms of GERD symptom alleviation and esophagitis. Many trials have been conducted to establish the beneficial effect of PPIs on asthma symptoms and pulmonary function tests. However, their advantage has not been clearly shown and remains obscure.
Treatment with antireflux therapy did not consistently improve any one measure of pulmonary function in patients with asthma, but some studies did report objective improvement in single measurements of lung function such as FEV1, morning PEF, and evening PEF. The variability in reporting of asthma symptoms in the included studies prevented detailed analysis of any parameter except for nocturnal asthma symptoms which were noted to be improved by some authors in patients treated with histamine antagonists.[7,17,18]
Kiljander and associates assessed the effects of esomeprazole 40 mg twice daily versus placebo for 16 weeks among 770 patients with moderate-to-severe asthma. The study included asthmatics with moderate-to-severe asthma based on FEV1 (50–80% predicted) and evidence of bronchodilator reversibility. The presence of GERD was based primarily on patient reported clinical symptoms.[19] They included 961 patients with moderate-to-severe asthma with demonstrated reversibility of airflow obstruction and ≥1 clinically important asthma exacerbation within the preceding 12 months with symptomatic GERD (as determined by the Reflux Disease Questionnaire. After 26 weeks of therapy, there was no improvement noted in the primary endpoint of the study, change from baseline mean morning PEF, in either study group. In addition, no differences were noted in use of rescue bronchodilators, percentage of asthma symptom-free days, or frequency of severe asthma exacerbations. Treatment with esomeprazole at both doses was associated with a statistically significant improvement in FEV1 and asthma-related quality of life throughout the study period. However, the improvement in FEV1 was small and likely of small clinical significance.[20]
Therefore, in some asthmatics with symptoms suggestive of GERD, the available literature suggests that there may be a small benefit of treatment of GERD on some aspects of asthma-related quality of life and possibly on exacerbations, but no clear evidence supports objective benefits in pulmonary function or overall asthma control. However, one could argue that treatment for symptomatic GERD is warranted in patients with asthma independent of any potential benefit on asthma outcomes if the underlying GERD warrants treatment on its own. The current NIH guidelines recommend an empiric trial of GERD therapy in poorly controlled asthmatics even if they do not have GERD symptoms.[21]
The American Lung Association Asthma Clinical Research Centers conducted a multicenter, randomized, and placebo-controlled trial of esomeprazole 40 mg twice daily for 24 weeks in patients with poorly controlled asthma and minimal or no symptoms of (silent) GER, using ambulatory esophageal pH monitoring to confirm the presence or absence of GER in participants. Four hundred and twelve patients on moderate or high doses of inhaled corticosteroids with inadequately controlled asthma (defined as either a Juniper Asthma Control Questionnaire of 1.5 or higher or the presence of more than one unscheduled health-care visit for asthma in the preceding 12 months) underwent pH testing and were randomly assigned to treatment with esomeprazole or placebo independent of pH study results. The diagnosis of asthma was based on physician diagnosis with either a positive methacholine challenge test or evidence of bronchodilator reversibility of the FEV1 with an increase of ≥12% after short-acting bronchodilator administration.[22,23] After 6 months of therapy, episodes of poor asthma control occurred with similar frequency in the placebo and esomeprazole groups (2.3 vs. 2.5 events/person-year respectively; P = 0.66). Although night-time awakenings due to asthma occurred on more than one occasion in over half of the subjects, there was no significant difference between groups. Similarly, there was no difference with respect to lung function (including PEF, FEV1, and methacholine responsiveness), asthma symptoms, or asthma related quality of life when patients treated with esomeprazole were compared with those who received placebo. Ambulatory pH monitoring demonstrated acidic GER in approximately 40% of both groups of participants, but failed to identify a subgroup of patients that was likely to benefit from therapy with the PPI. From these findings, Mastronarde et al. concluded that the empiric use of acid suppression in patients with poorly controlled asthma but minimal or no GERD symptoms should not be routinely recommended.[22]
The potential importance of the location of reflux in the esophagus (proximal vs. distal) has also been considered as a potential important factor in the clinical relationship between asthma and GERD. In a small study (n = 133) of subjects with upper airway symptoms attributed to GERD, dual-probe esophageal pH monitoring demonstrated a significantly higher incidence of nocturnal cough in those participants with acid reflux detected at both the proximal and distal probe than in those in whom reflux was detected at the distal probe alone. While the possibility that the participants’ nocturnal cough may have been related to uncontrolled asthma was not specifically addressed by the authors, the study still raises the important issue that symptoms from proximal GERD such as cough may mimic asthma and may confound measurements of asthma control in the clinical and research setting.[24] A subgroup (n = 242) of participants in the American Lung Association’s Study of acid reflux and asthma underwent dual-probe pH esophageal monitoring. Thirty-eight percent of participants demonstrated proximal GERD, but concordance between proximal and distal GERD on pH probe was poor.[22] While the presence of proximal GERD was associated with worse asthma-related quality of life, no differences were noted in other the asthma-related clinical outcomes measures described above. It has been hypothesized that nonacidic or alkali reflux may affect asthma control parameters, but, to date, this interesting theory remains untested.[25]
CONCLUSION
GERD and asthma show bidirectional association and shared common pathophysiological mechanisms. Reflex and reflux theories were associated with GERD and are linked with worsening of asthma and positive effects on asthma control have been globally reported with relief of GERD. Asthma medicines are linked with worsening of existing GERD and in proportionate number of cases with asymptomatic GERD become symptomatic ones.
At present, available data have shown limited benefits of treatment of symptomatic GERD on asthma outcomes and no clear benefit on asthma control. However, in a significant percentage of asthmatics, GERD is clinically silent or associated with only mild symptoms. Empiric treatment of clinically silent GERD in patients with uncontrolled asthma does not seem to be an effective therapy for asthma and is not supported by current data. Instead, the clinician should focus on other factors which can affect asthma control such as use of adequate controller medications, patient compliance, proper inhaler technique, and control of other significant comorbid conditions.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Natural history, pathophysiology and evaluation of gastroesophageal reflux disease. Dis Mon. 2020;66:100848.
- [CrossRef] [PubMed] [Google Scholar]
- Gastroesophageal reflux disease and asthma: A narrative review. Cureus. 2022;14:e24917.
- [CrossRef] [Google Scholar]
- Chronic cough due to gastroesophageal reflux in adults: CHEST guideline and expert panel report. Chest. 2016;150:1341-60.
- [CrossRef] [PubMed] [Google Scholar]
- Esophageal reflux as a trigger in asthma. Dis Chest. 1966;49:464-6.
- [CrossRef] [PubMed] [Google Scholar]
- The association between gastro-oesophageal reflux disease and asthma: A systematic review. Gut. 2007;56:1654-64.
- [CrossRef] [PubMed] [Google Scholar]
- Approach to the patient with presumed extraoesophageal GERD. Best Pract Res Clin Gastroenterol. 2013;27:415-31.
- [CrossRef] [PubMed] [Google Scholar]
- The relationship between gastroesophageal reflux and asthma: An update. Ther Adv Respir Dis. 2011;5:143-50.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of bronchial obstruction on lower esophageal sphincter motility and gastroesophageal reflux in patients with asthma. Am J Respir Crit Care Med. 2002;166:1206-11.
- [CrossRef] [PubMed] [Google Scholar]
- The effects of an inhaled beta(2)-adrenergic agonist on lower esophageal function: A dose-response study. Chest. 2001;120:1184-9.
- [CrossRef] [PubMed] [Google Scholar]
- Bronchial responsiveness during esophageal acid infusion. Lung. 2008;186:123-8.
- [CrossRef] [PubMed] [Google Scholar]
- Acid stress in the pathology of asthma. J Allergy Clin Immunol. 2004;113:610-9.
- [CrossRef] [PubMed] [Google Scholar]
- The spectrum of pulmonary symptoms due to gastroesophageal reflux. Thorac Surg Clin. 2005;15:353-68.
- [CrossRef] [PubMed] [Google Scholar]
- Muscarinic receptors involved in airway vascular leakage induced by experimental gastro-oesophageal reflux. Life Sci. 2008;82:949-55.
- [CrossRef] [PubMed] [Google Scholar]
- Extra-esophageal gastroesophageal reflux disease and asthma: Understanding this interplay. Expert Rev Gastroenterol Hepatol. 2015;9:969-82.
- [CrossRef] [PubMed] [Google Scholar]
- 24-h esophageal pH testing in asthmatics: Respiratory symptom correlation with esophageal acid events. Chest. 1999;115:654-9.
- [CrossRef] [PubMed] [Google Scholar]
- Gastroesophageal reflux disease and asthma exacerbation: A systematic review and meta-analysis. Pediatr Allergy Immunol. 2022;33:e13655.
- [CrossRef] [PubMed] [Google Scholar]
- Asthmatics with gastroesophageal reflux: Long term results of a randomized trial of medical and surgical antireflux therapies. Am J Gastroenterol. 2003;98:987-99.
- [CrossRef] [PubMed] [Google Scholar]
- A trial of ranitidine in asthmatic children and adolescents with or without pathological gastro-oesophageal reflux. Eur Respir J. 1992;5:201-6.
- [CrossRef] [PubMed] [Google Scholar]
- Effects of esomeprazole 40 mg twice daily on asthma: A randomized placebo-controlled trial. Am J Respir Crit Care Med. 2006;173:1091-7.
- [CrossRef] [PubMed] [Google Scholar]
- Effect of esomeprazole 40 mg once or twice daily on asthma: A randomized, placebo-controlled study. Am J Respir Crit Care Med. 2010;181:1042-8.
- [CrossRef] [PubMed] [Google Scholar]
- Expert Panel Report 3: Moving forward to improve asthma care. J Allergy Clin Immunol. 2007;120:1012-4.
- [CrossRef] [PubMed] [Google Scholar]
- Efficacy of esomeprazole for treatment of poorly controlled asthma. N Engl J Med. 2009;360:1487-99.
- [CrossRef] [PubMed] [Google Scholar]
- Identifying 'well-controlled' and 'not well-controlled' asthma using the Asthma Control Questionnaire. Respir Med. 2006;100:616-21.
- [CrossRef] [PubMed] [Google Scholar]
- Symptom predictability of reflux-induced respiratory disease. Dig Dis Sci. 2002;47:9-14.
- [CrossRef] [PubMed] [Google Scholar]
- Silent acid reflux and asthma control. N Engl J Med. 2009;360:1551-3.
- [CrossRef] [PubMed] [Google Scholar]








