Translate this page into:
The protective role of Vitamin E against copper-induced histopathological changes in rat rectum: An experimental study

*Corresponding author: Joseph Raymond Enoghase, Department of Anatomy, University of Benin, Benin City, Edo, Nigeria. enoghase41@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Enoghase JR, Obayuwana E, Osarinmwian BI. The protective role of Vitamin E against copper-induced histopathological changes in rat rectum: An experimental study. Glob J Health Sci Res. doi: 10.25259/GJHSR_21_2024
Abstract
Objectives
This study aims to evaluate the impact of Vitamin E supplementation on mitigating copper-induced histopathological changes in rat rectum tissues.
Material and Methods
Twenty-five Wistar rats were divided into five groups and subjected to different dosages of copper sulfate, Vitamin E, or a combination. Tissue samples were collected, processed, and stained with Hematoxylin and Eosin for histopathological examination.
Results
Histopathological analysis revealed significant alterations in the rat rectum following exposure to copper sulfate alone, including decreased cryptgoblet cells and mucosal lymphocyte mobilization. Vitamin E supplementation showed mitigated effects, with signs of active mucosal congestion and lymphocyte mobilization. However, when rats were administered copper sulfate followed by Vitamin E, histopathological changes were normalized, indicating a protective role for Vitamin E. Conversely, rats exposed to copper sulfate without subsequent Vitamin E supplementation exhibited severe inflammatory responses and submucosal vascular ulceration.
Conclusion
Vitamin E demonstrates a protective effect against copper-induced histopathological changes in rat rectum tissues, potentially mediated through its antioxidant properties. Further molecular studies are warranted to elucidate the underlying mechanisms. Hematoxylin and Eosin staining proved instrumental in identifying and characterizing these histopathological alterations.
Keywords
Antioxidant protection
Copper sulfate
Histopathological changes
Rat rectum
Vitamin E
INTRODUCTION
The importance of Vitamin E in maintaining various aspects of health cannot be overstated. It acts as a potent antioxidant, safeguarding cell membranes from oxidative damage caused by free radicals.[1] This protective function extends to immune support, heart health, skin maintenance, and eye health.[2] Vitamin E is primarily obtained through a diet rich in fruits, vegetables, nuts, and seeds.[3]
Research indicates that Vitamin E has a multifaceted role within the body, particularly as a fat-soluble antioxidant.[4] It neutralizes free radicals by donating hydrogen atoms, thus preventing oxidative damage to cells.[5] Moreover, it influences gene expression and enzyme activity regulation,[6] notably in the modulation of protein kinase C activity.[7]
Vitamin E’s impact on gene expression involves its antioxidant properties, which can affect signaling pathways and transcription factors.[7] This modulation of gene expression extends to hepatic functions, cholesterol homeostasis,[8] and age-related changes.[9] Long-term deficiency may lead to alterations in hepatic gene expression, affecting various metabolic processes.[10]
Vitamin E plays a vital role in maintaining overall health by acting as a powerful antioxidant and modulating gene expression and enzyme activity. Its deficiency is rare and is usually associated with fat digestion issues rather than dietary insufficiency.[11]
Copper (II) sulfate, also known as cupric sulfate, is a versatile compound widely used across various industries.[12] Its production involves different methods tailored to scale and purity requirements, including direct synthesis and bioleaching.[13] The compound is commonly employed in laboratories due to its high purity and convenience.[13] However, its environmental impact and toxicity, dose-dependent and primarily linked to its copper content, raise concerns.[14] High concentrations can harm aquatic life and pose health risks to humans through various exposure routes.[15] Acute exposure may result in severe symptoms, while chronic exposure, though rare, can lead to long-term health effects.[16] In ecosystems, copper sulfate can disrupt aquatic fauna and ecosystem balance, necessitating careful management in pesticide use to avoid ecological disturbances.[17,18]
The rectum is an integral part of the gastrointestinal tract, serving as the terminal section of the large intestine.[19] It plays a crucial role in the process of digestion by being the final stop before fecal matter is expelled from the body.[20]
The rectum’s primary role is to act as a temporary storage facility for feces, playing a key part in the defecation process.[21]
The rectum, being the final section of the large intestine, is susceptible to various forms of toxicity, which can manifest as inflammation, pain, bleeding, or more severe health conditions.[22]
MATERIAL AND METHODS
Experimental rats
Twenty-five Wistar rats of weight between 120 g and 170 g were used. The rats were given a 2-week acclimatization period before the administration method began. They were given free access to conventional rat feed and water. The research ethic committee’s guidelines for animal treatment at the University of Benin’s College of Medicine were espoused and fully implemented.
Experimental protocol
All administrations were done by gavage and lasted for 60 days [Table 1].[23]
| Groups | Dosage |
|---|---|
| Group A | Served as control and were fed with animal feed and water ad libitum. |
| Group B | Received 200 mg/kg of copper sulfate daily for 30 days. |
| Group C | Received 200 mg/kg of Vitamin E daily for 30 days.[23] |
| Group D | Received 200 mg/kg of copper sulfate daily for 30 days followed by 200 mg/kg of Vitamin E daily for 30 days[23] |
| Group E | Received 200 mg/kg of copper sulfate daily for 30 days followed by distilled water daily for 30 days. |
Copper sulfate and Vitamin E were administered using orogastric tube by following a modified method by Alflen et al.[24]
Tissue collection, processing and staining, histopathology
The rats were sacrificed and the rectum was taken at the end of the 30-day and 60-day study. The tissue was immediately sent to the University of Benin Teaching Hospital’s Chemical Pathology department. The rectum tissue was preserved for 24 h in 10% buffered formalin before being histologically processed and stained with Hematoxylin and Eosin (H&E) using standard procedures.[25] The sections obtained were examined and photomicrographs were taken using a Leica DM750 research microscope with an attached digital camera (Leica CC50). The tissues were photographed digitally at magnifications of ×100.
RESULTS
The histopathological analysis showed notable differences in the rat rectum among the treatment groups. The control group had a normal mucosal lining, crypts, submucosa, and muscularis propria. Rats exposed to copper sulfate alone had a significant decrease in crypt goblet cells and increased mucosal lymphocyte activity, indicating damage to the mucosal barrier. In contrast, those given only Vitamin E showed active mucosal congestion and lymphocyte mobilization, suggesting an immune response. Rats that received copper sulfate followed by Vitamin E had normal crypt goblet cells, mural vasodilatation, and active congestion, demonstrating Vitamin E’s protective and restorative effects. However, those given copper sulfate followed by distilled water developed severe mucosal inflammatory infiltrates and submucosal vascular ulceration, indicating that the tissue damage persisted and worsened without Vitamin E intervention.
Rat rectum. Control. Showing: Mucosa Epithelium (ME), crypts (Cr), submucosa (SM), and muscularis propria (MP): Hematoxylin and Eosin (H&E) ×100 [Figure 1].
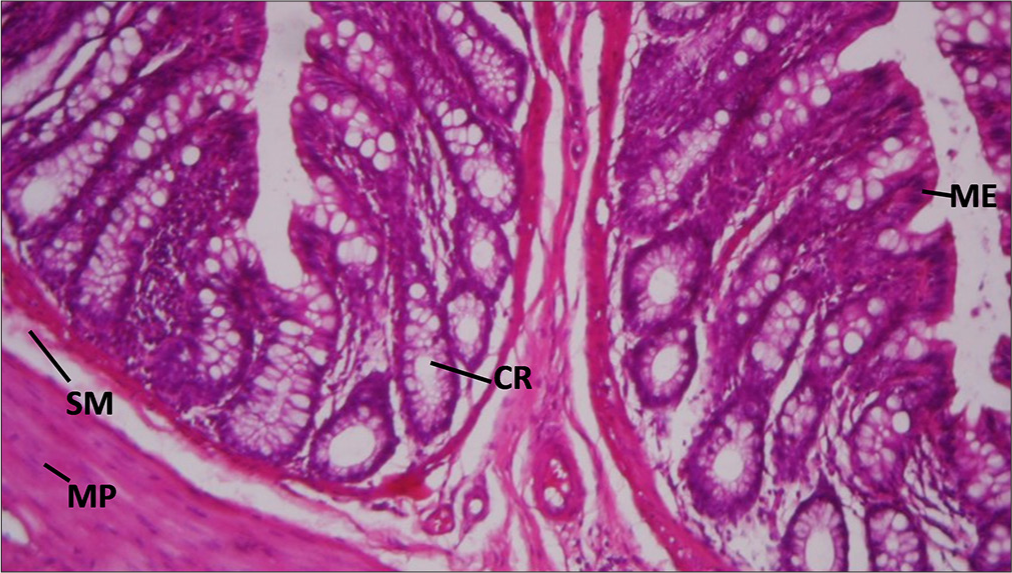
- Section of Rat’s rectum in control group. ME: Mucosa epithelium, CR: Crypts, SM: Submucosa, MP: Muscularis propria. Hematoxylin and Eosin ×100.
Rat rectum given copper only showing: Decreased Crypts goblet cells (GC) and mobilization of mucosal lymphocytes (ML): H&E ×100 [Figure 2].
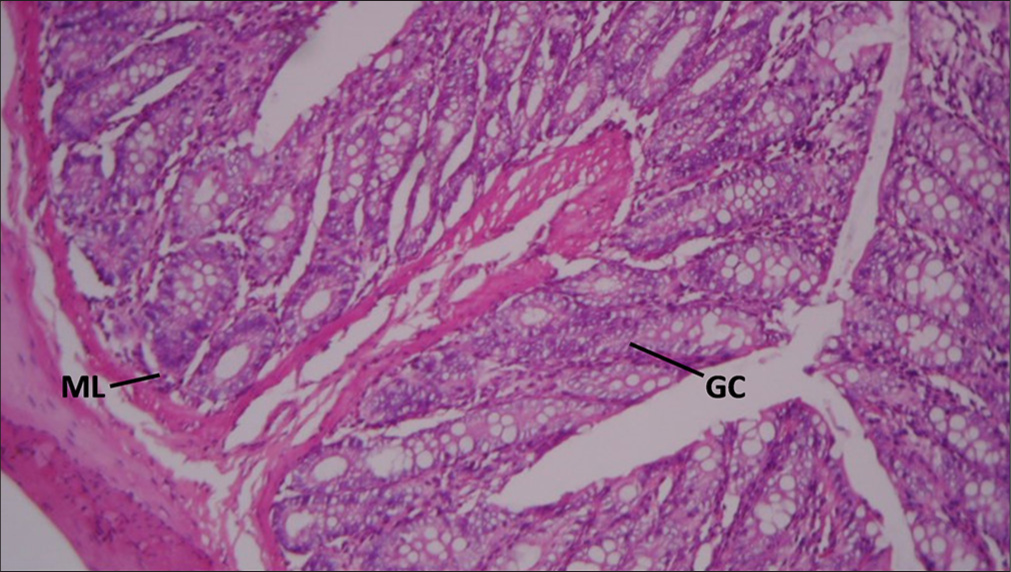
- Section of Rat's rectum administered 200mg/kg of copper sulfate only. GC: Goblet cells, ML: Mucosal lymphocytes. Hematoxylin and Eosin ×100.
Rat rectum given Vitamin E only showing: Active mucosal congestion (MC) and mobilization of Mucosa lymphocytes: H&E ×100 [Figure 3].
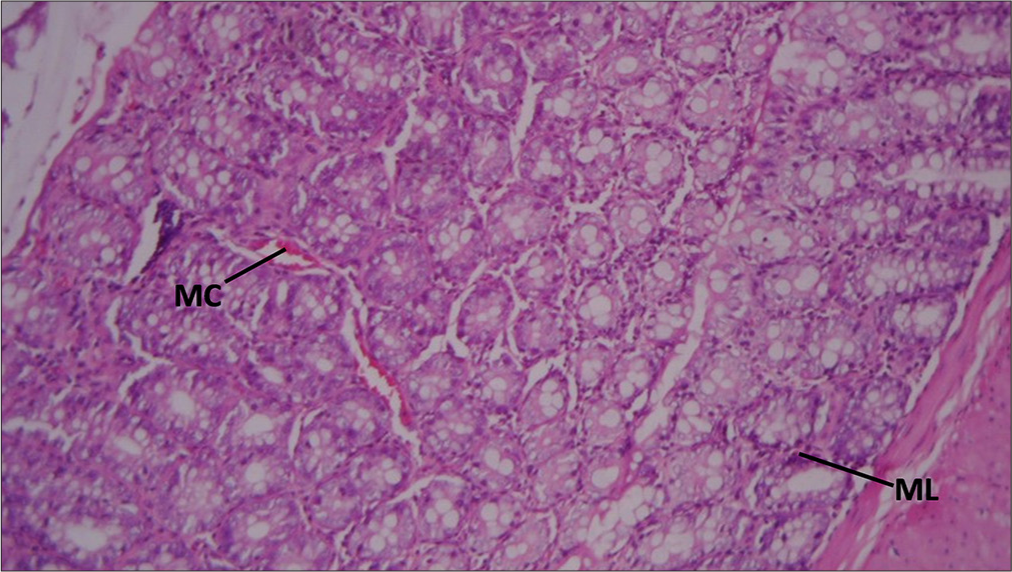
- Section of Rat's rectum administered with 200 mg/kg of Vitamin E only. MC: Mucosal congestion, ML: Mucosa lymphocytes. Hematoxylin and Eosin ×100.
Rat rectum given copper for 30 days, then Vitamin E for 30 days showing: Normal Crypts goblet cells and mural vasodilatation and active congestion (OC): H&E ×100 [Figure 4].
- Section of Rat's rectum administered with 200mg/kg of copper sulfate for 30 days then 200mg/kg of Vitamin E for 30 days. MI: Mucosal infiltrates, VU: Vascular ulceration. Hematoxylin and Eosin ×100.
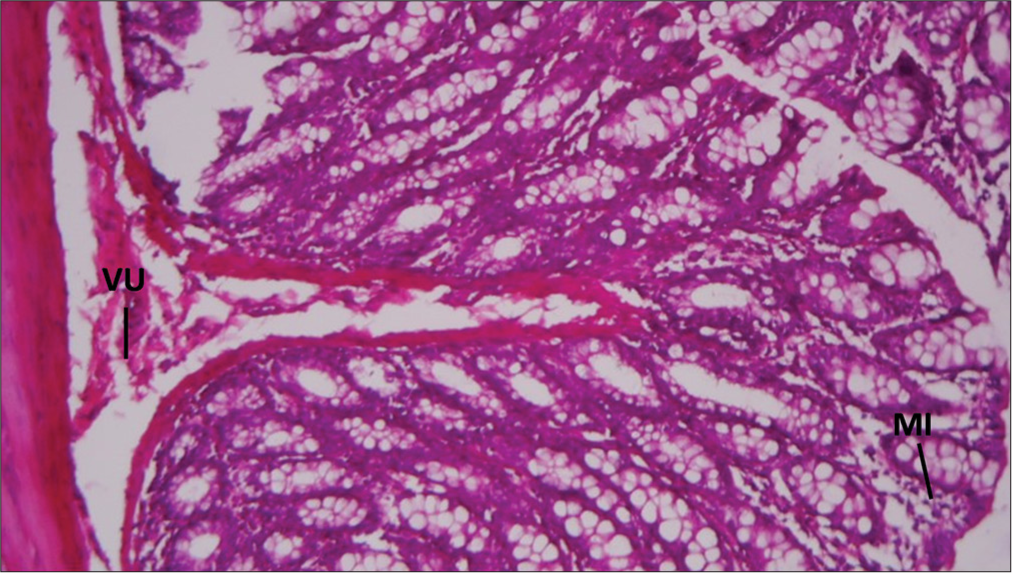
Rat rectum given copper for 30 days, then distilled water for 30 days, then distilled water for 30 days, showing mucosal infiltrates of inflammatory cells (MI) and submucosal Vascular ulceration (VU): H&E ×100 [Figure 5].
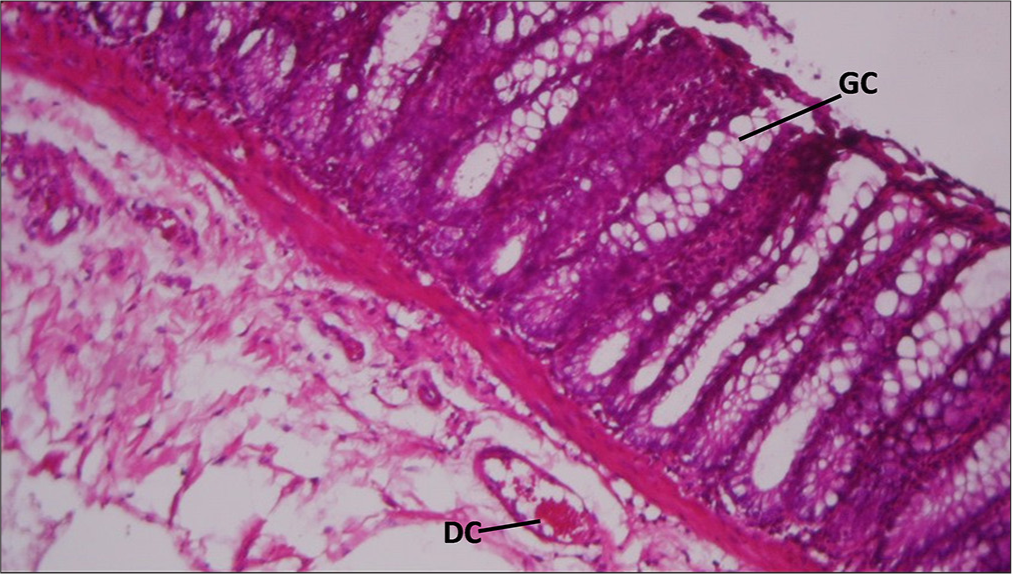
- Section of Rat's rectum administered with 200mg/kg of copper sulfate for 30 days then distilled water for 30 days. GC: Goblet Cells, DC: Mural vasodilatation and active congestion.
DISCUSSION
The control sample shows a normal histological structure with a mucosal lining (ME), Crs, submucosa (SM), and MP. This serves as a baseline for comparison with treated samples.
Exposure to copper resulted in a decrease in Cr GCs and mobilization of MLs. This suggests that copper may have a detrimental effect on the mucosal barrier, as GCs are responsible for mucus production which protects the intestinal lining.[26] The presence of lymphocytes indicates an immune response, possibly due to irritation or injury to the mucosa.[26]
Vitamin E treatment showed active MC and mobilization of MLs. Congestion could be due to increased blood flow or inflammation, while the presence of lymphocytes again suggests an immune response. Vitamin E is known for its antioxidant properties, which may explain the inflammatory response as the body’s attempt to repair tissue damage.[27]
When rats were given copper for 30 days followed by Vitamin E for another 30 days, the samples showed normal Cr GCs and mural vasodilatation with active congestion (OC). This indicates that Vitamin E may have a protective or restorative effect on the damage induced by copper, as evidenced by the normalization of GCs. Vasodilatation and congestion might reflect ongoing reparative processes.[28]
Rats exposed to copper for 30 days and then switched to distilled water for the same duration exhibited mucosal infiltrates of inflammatory cells (MI) and submucosal VU. This severe reaction suggests that without the intervention of Vitamin E, the damage caused by copper persists and may even worsen, leading to ulceration and significant inflammation.[29]
CONCLUSION
The results suggest that copper has a potentially harmful effect on the rat rectum, impairing the mucosal barrier and inducing an inflammatory response. Vitamin E seems to mitigate these effects, possibly due to its antioxidant properties that help in reducing oxidative stress and promoting healing. However, the exact mechanisms of these interactions would require further molecular studies to explain. The use of H&E staining has been crucial in identifying these histopathological changes, as it allows for the clear visualization of tissue structure and cellular components.
Ethical approval
The research ethic committee’s guidelines for animal treatment at the University of Benin’s College of Medicine were espoused and fully implemented. No ethical number was obtained.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship: Nil.
References
- Vitamin E as an antioxidant. Ankara Eczacilik Fakul Derg. 2003;32:243-67.
- [CrossRef] [Google Scholar]
- The role of vitamin e in human health and some diseases. Sultan Qaboos Univ Med J. 2014;14:e157-65.
- [Google Scholar]
- Vitamin E: Nutritional aspects. In: Molecular nutrition. United States: Academic Press; 2020. p. :447-85.
- [CrossRef] [Google Scholar]
- Vitamin E beyond its antioxidant label. Antioxidants. 2021;10:634.
- [CrossRef] [PubMed] [Google Scholar]
- Free radicals, antioxidants in disease and health. Int J Biomed Sci. 2008;4:89-96.
- [CrossRef] [PubMed] [Google Scholar]
- Vitamin E: Regulatory role on gene and protein expression and metabolomics profiles. IUBMB Life. 2019;71:442-55.
- [CrossRef] [PubMed] [Google Scholar]
- Vitamin E: Regulatory role on signal transduction. IUBMB Life. 2018;71:456-78.
- [CrossRef] [PubMed] [Google Scholar]
- Vitamin E: Regulatory role in the cardiovascular system. IUBMB Life. 2019;71:507-15.
- [CrossRef] [PubMed] [Google Scholar]
- Vitamin E-gene interactions in aging and inflammatory age-related diseases: Implications for treatment. A systematic review. Ageing Res Rev. 2014;14:81-101.
- [CrossRef] [PubMed] [Google Scholar]
- Gene regulatory activity of vitamin E. Berlin: Springer eBooks; 2019:81-98.
- [CrossRef] [Google Scholar]
- Vitamin E trafficking in neurologic health and disease. Annu Rev Nutr. 2013;33:87-103.
- [CrossRef] [PubMed] [Google Scholar]
- Copper dithiocarbamates: Coordination chemistry and applications in materials science, biosciences and beyond. Inorganics. 2021;9:70.
- [CrossRef] [Google Scholar]
- Copper leaching from primary sulfides: Options for biological and chemical extraction of copper. Hydrometallurgy. 2006;83:10-20.
- [CrossRef] [Google Scholar]
- High sensitivity of embryo-larval stage of the Mediterranean mussel, Mytilus galloprovincialis to metal pollution in combination with temperature increase. Mar Environ Res. 2016;122:59-66.
- [CrossRef] [PubMed] [Google Scholar]
- Acute copper sulfate poisoning resulting from dermal absorption. Am J Ind Med. 2018;61:783-8.
- [CrossRef] [PubMed] [Google Scholar]
- Copper environmental toxicology, recent advances, and future outlook: A review. Environ Sci Pollut Res. 2019;26:18003-16.
- [CrossRef] [PubMed] [Google Scholar]
- Ecotoxicology of metals-sources, transport, and effects on the ecosystem. Netherlands: Elsevier eBooks; 2022:593-627.
- [CrossRef] [Google Scholar]
- Classification and functions of enteroendocrine cells of the lower gastrointestinal tract. Int J Exp Pathol. 2011;92:219-31.
- [CrossRef] [PubMed] [Google Scholar]
- Pathophysiology of short bowel syndrome. JPEN J Parenter Enteral Nutr. 2014;38:14S-22.
- [CrossRef] [Google Scholar]
- The physiology of human defecation. Dig Dis Sci. 2012;57:1445-64.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic treatment-induced gastrointestinal toxicity: Incidence, clinical presentation and management. Ann Gastroenterol. 2012;25:106-18.
- [Google Scholar]
- Ameliorative effect of vitamin E on copper sulfate-induced liver damage in Wistar rats. J Exp Clin Anat. 2024;21:220-6.
- [CrossRef] [Google Scholar]
- Orogastric tube insertion using the new gastric tube guide: First experiences from a manikin study. BMC Anesthesiol. 2017;17:54.
- [CrossRef] [PubMed] [Google Scholar]
- Copper changes intestinal microbiota of the cecum and rectum in female mice by 16S rRNA gene sequencing. Biol Trace Elem Res. 2019;193:445-55.
- [CrossRef] [PubMed] [Google Scholar]
- Prevention of peritoneal adhesions by intraperitoneal administration of vitamin E: An experimental study in rats. Dis Colon Rectum. 2004;47:2157-61.
- [CrossRef] [PubMed] [Google Scholar]
- Using cell nuclei features to detect colon cancer tissue in hematoxylin and eosin stained slides. Cytometry A. 2017;91:785-93.
- [CrossRef] [PubMed] [Google Scholar]
- Comparison of the anti-inflammatory effects of vitamin E and vitamin D on a rat model of dextran sulfate sodium-induced ulcerative colitis. Exp Ther Med. 2023;25:98.
- [CrossRef] [PubMed] [Google Scholar]







